Table Of Content

When the category “foreign policy” was narrowed to a specific aspect – “the war on terrorism” – far more people chose it; only 33% chose domestic policy while 52% chose the war on terrorism. Surveyors may conduct pilot tests or focus groups in the early stages of questionnaire development in order to better understand how people think about an issue or comprehend a question. Pretesting a survey is an essential step in the questionnaire design process to evaluate how people respond to the overall questionnaire and specific questions, especially when questions are being introduced for the first time.
2 Three types of research designs
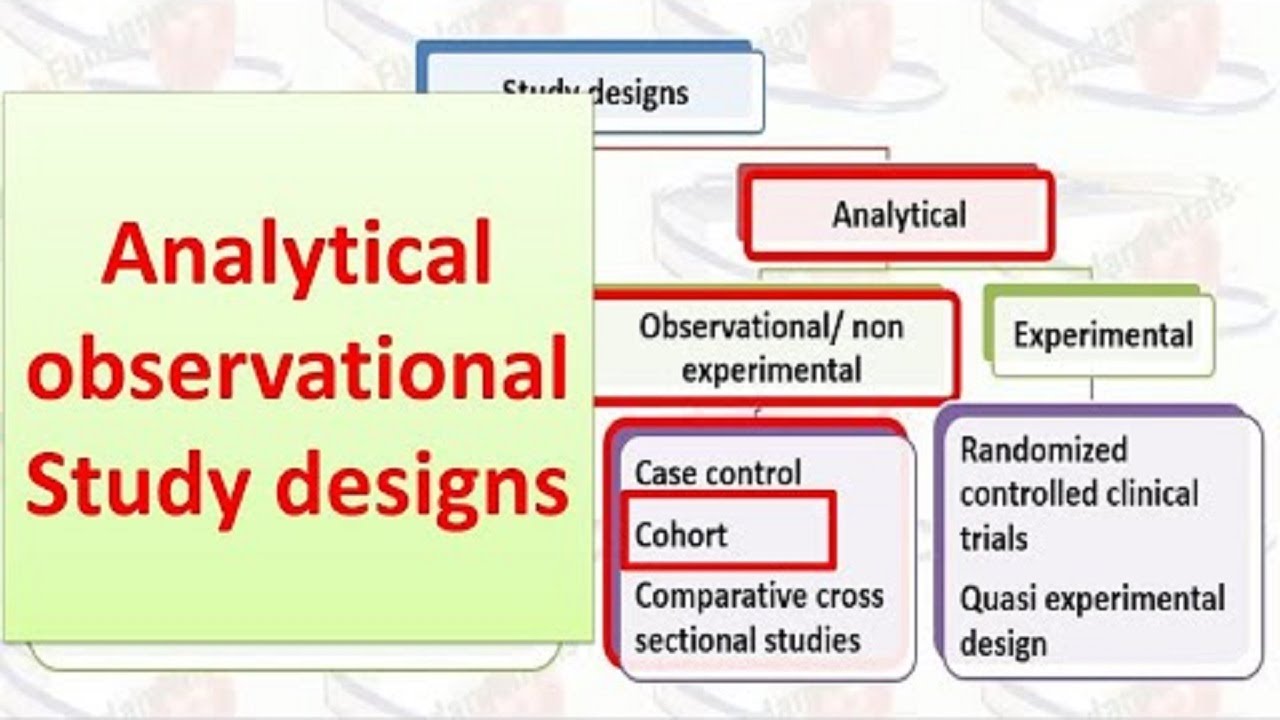
One of the main differences between a prospective cohort study and a randomized control trial is that instead of seeing the natural course of exposure (e.g., choice to smoke or not smoke), we instead randomly allocate participants into our study groups—we choose for them. This means we may give one group the standard of care for an ankle injury and give the other group a new cryotherapy plus standard of care to see the effect the cryotherapy has on the outcome of the injury. Observational study designs, also called epidemiologic study designs, are often retrospective and are used to assess potential causation in exposure-outcome relationships and therefore influence preventive methods. Observational study designs include ecological designs, cross sectional, case-control, case-crossover, retrospective and prospective cohorts.
OTHER RESEARCH METHODS
Because both exposure and outcome are assessed at the same time, temporality cannot be demonstrated, i.e. it cannot be demonstrated that the exposure preceded the disease (1–3,5,8). Point prevalence and period prevalence can be calculated in cross-sectional studies. Measures of risk for the exposure-outcome relationship that can be calculated in cross-sectional study design are odds ratio, prevalence odds ratio, prevalence ratio, and prevalence difference. Cross-sectional studies are relatively inexpensive and have data collected on an individual which allows for more complete control for confounding. Additionally, cross-sectional studies allow for multiple outcomes to be assessed simultaneously. In order to establish causality, it is important to be able to establish a temporal—or time—relationship between factors.
Types of Study Designs
In experimental studies, the researcher manipulates the exposure, that is he or she allocates subjects to the intervention or exposure group. Experimental studies, or randomised controlled trials (RCTs), are similar to experiments in other areas of science. That is, subjects are allocated to two or more groups to receive an intervention or exposure and then followed up under carefully controlled conditions. Such studies controlled trials, particularly if randomised and blinded, have the potential to control for most of the biases that can occur in scientific studies but whether this actually occurs depends on the quality of the study design and implementation. Some of the biases observed with cohort studies include selection bias and information bias.
Study designs: Part 1 – An overview and classification
On the other hand, qualitative studies can help researchers gain insights and generate testable hypotheses. They can be used parallel to quantitative studies to explore patients’ feelings, attitudes towards a new treatment, and personal tactics to cope with a disease. Assimilation effects occur when responses to two questions are more consistent or closer together because of their placement in the questionnaire. People were more likely to say that Republican leaders should work with Obama when the question was preceded by the one asking what Democratic leaders should do in working with Republican leaders (81% vs. 66%).
Developmental Psychology Research Methods - Verywell Mind
Developmental Psychology Research Methods.
Posted: Wed, 01 Nov 2023 07:00:00 GMT [source]
Table 1
The study design used to answer a particular research question depends on the nature of the question and the availability of resources. In this article, which is the first part of a series on "study designs," we provide an overview of research study designs and their classification. Primary research relies upon data gathered from original research expressly for that purpose (1,3,5). Secondary research focuses on single or multiple data sources that are not collected for a single research purpose (14,15).
Non-randomized trial study design
The relationship of an attribute to the disease is examined by comparing diseased and non-diseased persons with regard to the frequency or levels of the attribute in each group. Test-tube Lab Research "Test tube" experiments conducted in a controlled laboratory setting. This is a study in which 1) There are two groups, one treatment group and one control group. The treatment group receives the treatment under investigation, and the control group receives either no treatment (placebo) or standard treatment.
Institutional Review Board – Application Sample...
They will then develop closed-ended questions based off that pilot study that include the most common responses as answer choices. In this way, the questions may better reflect what the public is thinking, how they view a particular issue, or bring certain issues to light that the researchers may not have been aware of. Many surveyors want to track changes over time in people’s attitudes, opinions and behaviors.
Explaining the many kinds of research studies - Fred Hutchinson Cancer Center
Explaining the many kinds of research studies.
Posted: Thu, 13 Feb 2020 08:00:00 GMT [source]
Question wording
Also, since case-control studies do not begin with a population at risk, it is not possible to determine the true risk of outcome. Instead, one can only calculate the odds of association between exposure and outcome. Based on the direction of inquiry, study designs may be classified as forward-direction or backward-direction.
Reproducibility of Results - The statistical reproducibility of measurements (often in a clinical context), including the testing of instrumentation or techniques to obtain reproducible results. The concept includes reproducibility of physiological measurements, which may be used to develop rules to assess probability or prognosis, or response to a stimulus; reproducibility of occurrence of a condition; and reproducibility of experimental results. This compares a population with a certain medical condition with another group of people who do not have the disease but are otherwise similar to the study population. As an example, an ethnographer may study how different communities celebrate traditional festivals or how individuals from different generations interact with technology differently. This may involve a lengthy period of observation, combined with in-depth interviews to further explore specific areas of interest that emerge as a result of the observations that the researcher has made. Clinical trials are further divided into randomized clinical trial, non‐randomized clinical trial, cross‐over clinical trial and factorial clinical trial.
This short article gives a brief guide to the different study types and a comparison of the advantages and disadvantages. Other studies (Yap et al. 2021) suggest that people on the autism spectrum are more likely to be 'picky eaters', which contributes to the differences in gut bacteria. Quasi-experiments are similar to true experiments (i.e., answer relational RQs) where treatments are allocated to groups that already exist (e.g., may be naturally occurring). Methodological studies, just like pilot studies, are also paramount in research. They are studies that aim to test if research methods are accurate and repeatable (Peat, 2011).
Of these, demonstrating temporality is the only mandatory criterion for suggesting temporality. Therefore, prospective studies that follow study participants forward through time, including prospective cohort studies and interventional studies, are best suited for suggesting causation. Additionally, causation between an exposure and an outcome cannot be proven by one study alone; multiple studies across different populations should be considered when making causation assessments (17). Interventional studies are often prospective and are specifically tailored to evaluate direct impacts of treatment or preventive measures on disease. Each study design has specific outcome measures that rely on the type and quality of data utilized. Additionally, each study design has potential limitations that are more severe and need to be addressed in the design phase of the study.
Just like other trials with a preference group, comprehensive cohort studies do not provide definitive information about the effectiveness and the efficacy of a treatment, but supplemental results. Therefore, a further independent randomized controlled trial will be needed to provide clear evidence and to answer generalizability goals. Just like with randomized trials, they involve the procedure of researchers allocating subjects randomly. However, the difference here is that subjects receive two or more treatments one after another; so, the randomization refers to the order patients receive each treatment (Peat, 2011). In other words, this design is a repeated measurements design, and subjects cross over treatments during the study. In comparison, in parallel studies subjects stay on the same treatment during the whole trial.
The animation below compares observational, quasi-experimental and true experimental designs. Definition 3.4 (Experiment) Experimental studies (or experiments) study relationships with an intervention. As such, case reports provide information about one patient or a small group of patients, and case series – about larger groups of patients.
The essential feature is that some of the persons under study have the disease or outcome of interest and their characteristics are compared with those of unaffected persons. Random Allocation - A process involving chance used in therapeutic trials or other research endeavor for allocating experimental subjects, human or animal, between treatment and control groups, or among treatment groups. Logistic Models - Statistical models which describe the relationship between a qualitative dependent variable (that is, one which can take only certain discrete values, such as the presence or absence of a disease) and an independent variable.

No comments:
Post a Comment